Final Call: Unique MDNA55 Glioblastoma Trial Closing Soon
by Zara Jethani
Pacific Neuroscience Institute, Nationwide Site Leader of the MDNA55 Clinical Trial, Offers Final Enrollment for Glioblastoma Patients
Santa Monica, CA, February 4, 2019 – The Pacific Brain Tumor Center at Pacific Neuroscience Institute, in collaboration with the Saint John’s Cancer Institute at Providence Saint John’s Health Center, is the leading enrollment site for the MDNA55 clinical trial from Medicenna Therapeutics. This study offers an innovative treatment option using a new minimally invasive technique to deliver potent anti-tumor therapy directly into the tumor of patients with glioblastoma, an aggressive and lethal form of brain cancer.
Now in the final stages of recruitment, the clinical trial is currently still open to eligible patients.
The study registered its first patient in April 2017 and is scheduled to complete enrollment in the next couple of months. Patients receive treatment and follow-up care from PNI’s team of compassionate and experienced specialists.
The open-label, non-randomized, multi-center phase-2 study assesses safety and efficacy of convection-enhanced delivery (CED) of a new bioengineered targeted therapeutic, MDNA55, in adults with recurrent or progressive glioblastoma.
For patients who are no longer responding to first line chemotherapy treatment after surgery, MDNA55 is directly infused into the brain at the site of the tumor. This form of direct infusion, using targeted CED therapy, is of significant scientific interest as a method for achieving better drug delivery and efficacy to target tumor cells while reducing side effects associated with systemic and generalized delivery methods typical of standard therapies, like chemotherapy or radiation.

Lead Principal Investigators, Achal Singh Achrol, MD, and Santosh Kesari, MD, PhD, are encouraged by this trial and the method of bypassing the blood brain barrier (BBB) to optimize concentrations of therapeutic agent within the tumor. “Systemic treatments do not cross the BBB well, so not enough anti-tumor agent can get through to the brain tumor to be effective,” said neuro-oncologist Dr. Kesari. “With this precision approach, we can better target the tumor and surrounding areas directly. Recurrent glioblastoma has been shown to occur within centimeters of the original site, so we’re hoping this approach will destroy the active cancer cells that remain after the initial surgery, to minimize recurrence.”
“Because overall survival of glioblastoma patients after diagnosis with standard therapy alone is on average only a year in many community-based studies, there is significant importance to clinical trials in developing new therapeutic approaches for patients with glioblastoma,” neurosurgeon Dr. Achrol emphasized.
The process of CED in this trial involves mapping the patient’s tumor through specialized computer software and 3D imaging to determine optimal therapeutic coverage of the entire tumor and surrounding brain regions in preparation for the procedure. Dr. Achrol explained, “Because the therapy is delivered by micro-catheters, only very small openings of a few millimeters are required. We use real-time MRI to monitor the MDNA55 delivery after the tiny catheters have been placed along computationally-planned trajectories.”
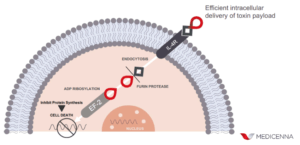
“MDNA55 is bioengineered to act like a ‘trojan horse’ delivery system. It does this by binding to specific cell receptors, called interleukin-4 receptors, found on tumor cells but not normal brain tissue. Once attached to these targeted cells, the protein delivers a toxic payload that kills only the target cells while leaving surrounding healthy tissue unharmed,” he added.
While chemotherapy only kills rapidly-dividing cancer cells at specific stages in the cell cycle, MDNA55 is unique in that its mechanism of action does not depend on rate of cell division or cell cycle stage. Its targeted payload can therefore kill both rapidly-dividing and slow-growing cancer cells, such as cancer stem cells, thus increasing the odds that more of the cancer will be destroyed. In addition, immune-suppressing cells of the tumor micro-environment (TME), which act like a cloak that hides cancer cells from our immune system, also express the IL4 receptors. Therefore, in addition to targeting tumor cells directly, MDNA55 has the potential to also cleanse the TME of enabling immunosuppressive cells to better expose cancer cells to tumor-killing immune responses.
Fahar Merchant, PhD, President and CEO of Medicenna Therapeutics, stated, “We are very fortunate to have the impressive expertise of Drs. Achrol and Kesari at the Pacific Neuroscience Institute in this important clinical trial. We are especially encouraged with promising survival results in the ongoing clinical trial following a single infusion of MDNA55 at the low dose. With patient recruitment at the high dose nearing completion, we expect better patient outcomes as demonstrated by earlier clinical and pre-clinical studies.”
Pacific Neuroscience Institute is one of only seven sites across the country that offers this clinical trial.
What is Glioblastoma?
Glioblastoma (also called glioblastoma multiforme or GBM) is classified as a Grade 4 astrocytoma and represents the most common and aggressive malignant primary brain tumor. Patients experience a multitude of symptoms including headaches, nausea, generalized changes in clarity of thought and attention, vision changes, weakness, numbness, or seizures. Standard glioblastoma treatment typically involves neurosurgical resection followed by chemotherapy and radiotherapy. Due to the invasive nature of the tumor, complete removal of the entire tumor is not usually feasible and unfortunately recurrence is common.
MDNA55 Trial Enrollment
This trial remains open to enrollment, and there are numerous other advanced therapy options available at Pacific Neuroscience Institute and Saint John’s Cancer Institute. Patients with gliomas, glioblastomas and other malignant brain tumors are encouraged to contact us to find out more.
Adults 18 years and older with recurrent or progressive glioblastoma are invited to participate in the MDNA55 trial.
For more information and to enroll into the trial, please contact us at 310-582-7437.
Full trial information | ClincalTrials.gov
Story adapted from original press release, February 4, 2019.
About the Author

Zara Jethani
Zara is the marketing director at Pacific Neuroscience Institute. Her background is in molecular genetics research and healthcare marketing. In addition, she is a graphic designer with more than 20 years experience in the healthcare, education and entertainment industries.
Last updated: June 10th, 2023