The Many Manifestations of Meningiomas
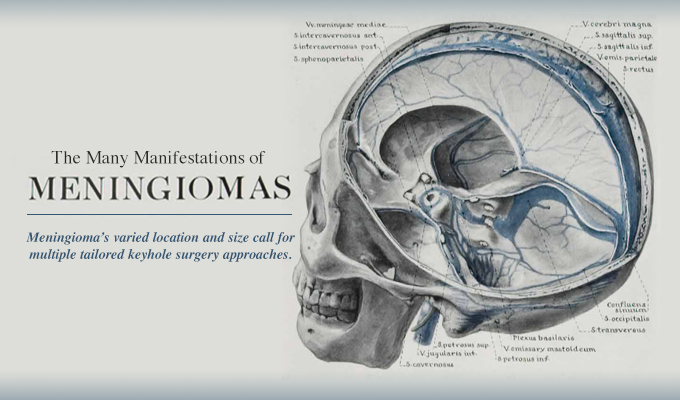
Eighty years ago, the father of modern neurosurgery, Harvey Cushing, published a now-famous monograph on his more than 30-year experience treating more than 300 meningiomas beginning in 1896. Back then, safely removing such formidable tumors without a surgical microscope, or the endoscope and modern anesthetic techniques was extremely challenging and risky. Nonetheless, Cushing’s surgical skills, attention to detail and perseverance paid off for his patients and the future of neurosurgery.

Flash forward to the present, and meningioma surgery has truly evolved into a nuanced array of surgical procedures tailored to the specifics of the tumor location and size. Given that this most common primary brain tumor can arise literally anywhere within the intracranial dural surfaces – the thick membrane that surrounds the brain – and even inside the fluid filled chambers (ventricles) of the brain, neurosurgeons need to have a full surgical armamentarium for their removal, ideally using keyhole techniques that maximize tumor removal and minimize risk of collateral damage and complications.
Our comprehensive team approach for patients with meningiomas has evolved dramatically over the years and overall outcomes, as one would hope, are much improved since the early days of neurosurgery. Herein we provide an overview of meningiomas, their symptoms, diagnosis and minimally invasive treatment options.
OVERVIEW
Meningiomas can occur at any age but are most common in adult women. They arise from the arachnoid (one of the coverings of the brain) and most (85-90%) are slow-growing benign tumors, pathologically defined as “typical meningiomas”, while 10-12% are “atypical meningiomas” (more aggressive) and approximately 1% are “anaplastic meningiomas” (very aggressive). They are usually not discovered until they impinge upon important brain structures resulting in symptoms, although these days, given that so many patients get an MRI or CT scan for a headache, dizziness or other complaints, many meningiomas are discovered “incidentally”. First-line treatment for symptomatic meningiomas is maximal safe surgical removal. Second-line treatment is typically focused radiotherapy or radiosurgery.
SYMPTOMS
Symptoms of meningioma depend entirely upon their location. For example:
- Meningiomas that arise close to the optic nerves or chiasm cause progressive vision loss.
- Meningiomas that arise in the skull base region of the cavernous sinus or Meckel’s cave where the nerves that control eye movements and facial sensation, can cause double vision or facial pain and numbness.
- Those that arise along the skull base region behind the ear in the area called the cerebellopontine angle can cause facial weakness, loss of hearing, difficulty swallowing and incoordination.
- One of the most common subsets of meningiomas so-called “convexity meningiomas” arise from the dural surface of the skull over the top or along the side of the brain, and typically become symptomatic when they become quite large (over 3 or 4 cm in diameter). These meningiomas put progressive pressure on the underlying brain, causing location-dependent symptoms such as personality changes, seizures, headaches, motor weakness in the arms or legs, incoordination, loss of sensation, spatial disorientation and visual field deficits.
DIAGNOSIS & ADDITIONAL TESTING
Meningiomas are ideally diagnosed and characterized by MRI but a CT scan can also detect most meningiomas. They typically have a characteristic growth pattern and shape and are usually readily distinguishable from other common brain tumors such as gliomas, schwannomas, pituitary tumors and metastatic brain tumors. They are usually solid and may have calcifications within the tumor; they tend to brightly “enhance” (light-up) after a gadolinium-MRI and after iodinated contrast during a CAT scan.
For meningiomas that arise in specific locations, additional testing is often needed in addition to an MRI.
- For tumors that arise near the optic nerves and chiasm or are associated with vision loss or double vision, formal visual field and acuity testing as well as assessment by a neuro-ophthalmologist is needed.
- For tumors that arise in the region of the sella turcica, complete pituitary hormonal testing is needed.
- For tumors that arise near the internal auditory canal with pressure on the cranial nerves responsible for hearing, an audiogram is needed.
PNI’S EXPERIENCE IN KEYHOLE REMOVAL OF MENINGIOMAS
Over the last decade, Dr. Daniel Kelly, Garni Barkhoudarian and their PNI colleagues have collectively operated on over 300 patients with meningiomas and along the way have published our results for various subsets of these tumors, particularly for those in who we have used an endonasal or supraorbital keyhole approach. Over 20% of our meningioma surgeries are for patients who have a residual or recurrent meningioma, treated previously at outside institutions.
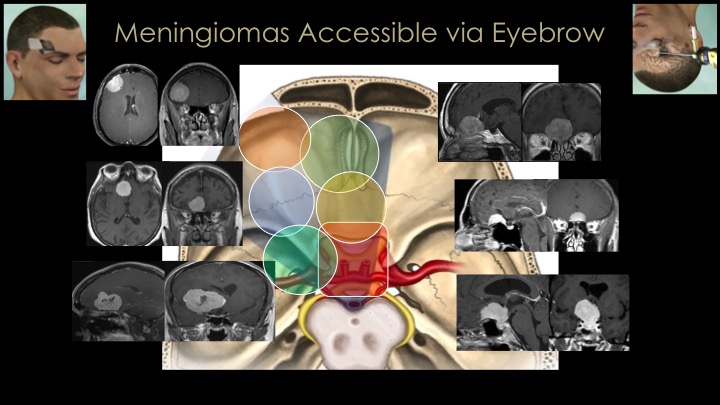
Because of advances in endoscopy, navigation and anatomical understanding, we now approach most meningiomas via a keyhole approach such as the supraorbital, minipterional, retromastoid or endonasal routes. For all of our meningioma surgeries, we emphasize use of surgical navigation (GPS for surgery), the Doppler probe for real-time artery and sinus localization to minimize vascular injury and stroke and to maximize surgical success and cranial nerve monitoring when needed.
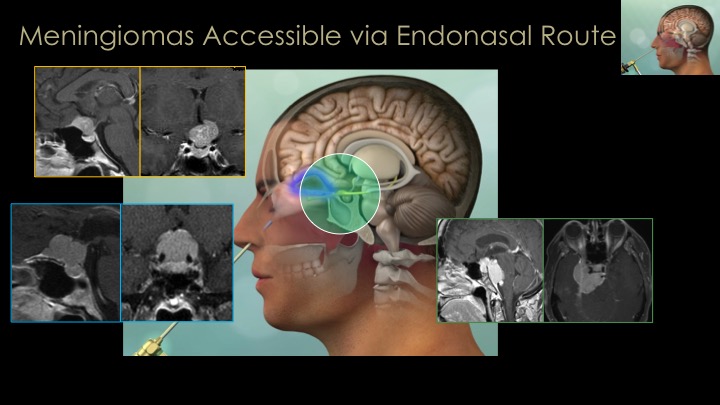
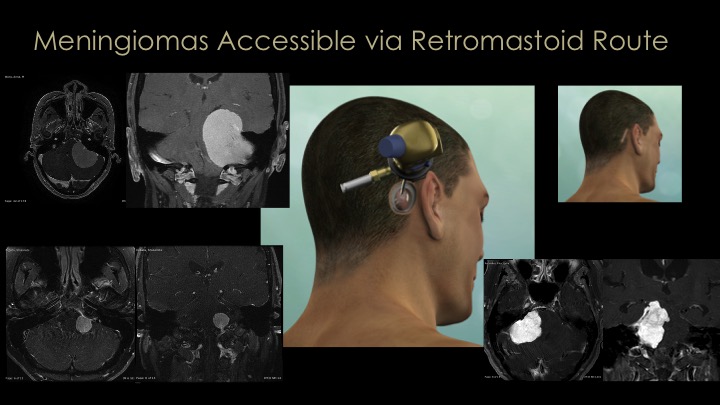
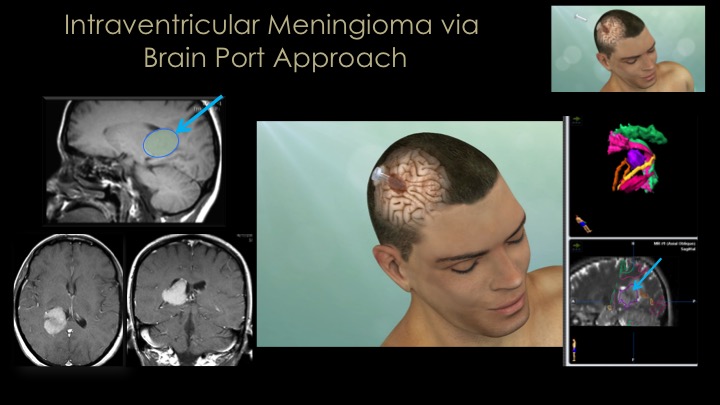
The great majority of meningiomas that arise around the midline skull base area in the region of the pituitary gland and optic nerves (so-called parasellar meningiomas), we remove via the nose (endonasal route) or through a supraorbital eyebrow incision. For large convexity meningiomas, a craniotomy strategically placed directly over the tumor is needed for complete removal. It is important to note that a substantial minority of meningiomas, particularly those of the skull base region are invasive into surrounding structures which precludes safe total removal. Incomplete removal is usually a result of tumor that is invasive into dural sinuses or compartments such as the cavernous sinus, Meckel’s cave or the sagittal sinus, or because the tumor is too adherent to or encasing critical brain arteries or cranial nerves or is simply too stuck to essential brain areas such as the motor cortex or brain stem. In such cases, small or even large remnants of tumor may have to be left behind and monitored with regular MRIs and then treated with focused radiation if they grow. Several examples of these approaches for meningiomas are shown here and in our meningioma video library.
Most patients undergoing craniotomy or endonasal removal of a meningioma are in the hospital for two nights and similarly, most patients undergoing endonasal meningioma removal are discharged home on day 2 after surgery.
FOLLOW-UP
Given that most meningiomas are benign and slow growing, patients need long-term neurosurgical follow-up with regular MRIs. Even patients with what appear to have complete tumor removal, early and late recurrences can occur. We typically follow our patients with regular MRIs every 6 months for at least 2 years post-surgery and then annually for at least 10 years. If a tumor regrowth does occur, options include repeat surgery, focused radiation (or radiosurgery) and in some instances targeted therapies or chemotherapy, or clinical trials.
Unfortunately, at this time, there is no consistently effective chemotherapy for meningiomas. However, for recurrent typical, atypical and anaplastic meningiomas, we routinely perform tumor genetic profiling to assess for therapeutic targets. Our PNI neuro-oncology team led by Dr. Santosh Kesari are actively investigating novel treatments for recurrent and previously treated meningiomas. Hopefully new molecular therapies are on the horizon for such aggressive meningiomas.
MENINGIOMA TEAM
Neurosurgery
[em-people id=”484″][em-people id=”645″][em-people id=”6612″]
ENT
[em-people id=”650″][em-people id=”653″]
Neuro-oncology
[em-people id=”648″][em-people id=”8424″]
Endocrinology
[em-people id=”660″][em-people id=”4821″]
Neuro-ophthalmology
[em-people id=”669″]
Radiation Oncology
[em-people id=”667″][em-people id=”665″]
For more about meningioma surgery or to get a second opinion about a newly diagnosed or recurrent meningioma, and to learn about our expert team, visit our meningioma page on the Pacific Brain Tumor Center website where you can schedule a consultation.

Daniel F. Kelly, MD, is the Director of the Pacific Neuroscience Institute in Santa Monica, CA. Considered to be one of the top neurosurgeons in the US, Dr. Kelly is internationally recognized in the field of minimally invasive keyhole surgery for brain, pituitary and skull base tumors. He continues to focus his efforts on advancing innovative treatments for patients, providing fellowship training in minimally invasive neurosurgery, and patient education and support.

Garni Barkhoudarian, MD, is a neurosurgeon with a focus on skull base and minimally invasive endoscopic surgery. He has particular interest and expertise in pituitary and parasellar tumors, brain tumors, skull-base tumors, intra-ventricular brain tumors, colloid cysts, trigeminal neuralgia and other vascular compression syndromes. He is an investigator in a number of clinical trials evaluating the efficacy of various medical or chemotherapies for pituitary tumors and malignant brain tumors. For virtually all tumors and intracranial procedures, Dr. Barkhoudarian applies the keyhole concept of minimizing collateral damage to the brain and its supporting structures using advanced neuroimaging and neuro-navigation techniques along with endoscopy to improve targeting and lesion visualization.

