

Updates on Management of Parkinson’s Disease
by Melita Petrossian
Changes in medical options for Parkinson’s disease
Advances in management of motor symptoms of Parkinson’s disease (PD) have been seen in recent years in both medical management and surgical management. However, both PD patients and their physicians may not be familiar with the increasing options and therefore may not be taking advantage of available treatments.
Many PD patients, after an initial period of stability, may develop motor complications, including dyskinesias (abnormal involuntary movements) and “on”/ “off” fluctuations. The latter refers to periods of time where the dopaminergic therapy (typically carbidopa/levodopa) is not reliably producing reductions in tremor, bradykinesia or rigidity. “Off” time may occur if the dose takes longer to kick in, the dose does not last long enough, or the dose does not seem to kick in at all, or stops working at unpredictable times. “Off” time may manifest as rigidity, bradykinesia, akinesia, freezing, dystonia, or tremor, but there is growing recognition of non-motor “off” symptoms, which may include anxiety, cognitive dulling, fatigue, respiratory fluctuations, and dizziness.
Patients experiencing bothersome dyskinesias and/or “on”/ “off” fluctuations are good candidates for advanced management of PD. This should begin with consultation with a movement disorders specialist and evaluation of the medication regimen. Because delayed gastric emptying and slow transit function of the gut are extremely common in PD patients, peripheral contributions to delayed “on” time play a large role. Thus, making sure that the medications are taken on an empty stomach, minimizing gut-slowing agents such as anti-cholinergics, and aggressively treating constipation are important first steps.
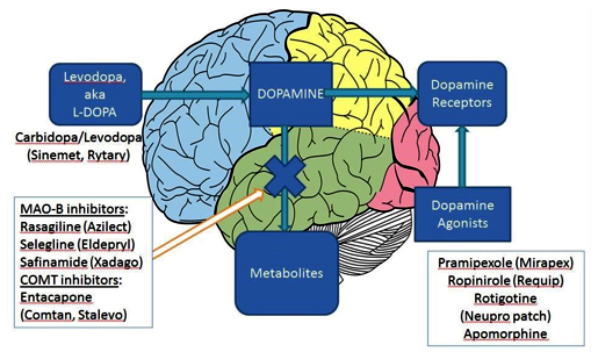
Medication adjustment may include increasing the frequency of carbidopa/levodopa, but for sensitive individuals this can increase dyskinesias. For many patients, taking carbidopa/levodopa more frequently evolves into taking medications six, seven, or even eight times a day, which becomes cumbersome. Another option is switching to a long-acting version of carbidopa/levodopa. Rytary is a capsule containing micro-beads that provide immediate and delayed release of carbidopa/levodopa, and is shown to reduce “off” time by about an hour per day. It is important to note that Rytary and immediate release carbidopa/levodopa are not dose-equivalent and there is a dose conversion of about 1.5-2x the dose. Therefore, if a PD patient is hospitalized it’s important to note that Rytary doses cannot be interchanged with immediate release carbidopa/levodopa doses.
Addition of an MAO-B inhibitor such as selegiline (Eldeprys), rasagiline (Azilect), or safinamide (Xadago), can improve “off” time, also by about an hour per day. These medications reduce metabolism of dopamine, both endogenous and that converted from exogenous levodopa. Safinamide is the newest of the three and was FDA approved in March 2017 for adjunct therapy with levodopa. Rasagiline is now available as a generic and is FDA approved both for adjunct therapy and monotherapy for mild parkinsonism. For patients who are taking antidepressants, MAO-B inhibitors prompt a drug interaction warning, mainly because MAO-B inhibitors are in the same class as MAO-A inhibitors. However, at approved doses, rasagiline has negligible MAO-A inhibitory effect, and safinamide is even more selective for the MAO-B enzyme. Therefore, when used with one antidepressant at approved doses, the risk of serotonin syndrome is extremely low. Serotonin syndrome may occur if MAO-B inhibitors are combined with more than one serotonergic substance (including Saint John’s wort, triptans, antidepressants, etc.), and patients should be advised to avoid cough/cold medication which contains dextromethorphan. Similarly, the tyramine crisis risk is negligible, and patients do not require any dietary modifications.
Addition of a COMT inhibitor such as entacapone, or exchanging carbidopa/levodopa with Stalevo (carbidopa/ levodopa/entacapone), can reduce “off” time by about an hour and a half. However, this approach may increase dyskinesias.
For those patients who have bothersome dyskinesias, amantadine has long been used off label to reduce dyskinesias. In August 2017, the FDA approved an extended-release form of amantadine, called Gocovri, which reduced dyskinesias by about 30% and improved off time by about an hour. While reports touted this as the “first” medication for treatment of dyskinesias, its mechanism of action and listed side effects are the same as the immediate release amantadine. Gocovri and immediate release amantadine have not been compared head-to-head in regards to efficacy and tolerability.
Consultation with a movement disorders specialist is a crucial step in navigating the changing landscape of medication options for PD.
Useful links:
Parkinson’s disease medications for motor symptoms
Parkinson’s disease medications for non-motor symptoms
Original article:
PhysicianLink – Your Link to Specialty Healthcare Providers – PLCMMC San Pedro and Torrance
About the Author
 Dr. Melita Petrossian is a Harvard-trained Neurologist and Movement Disorders specialist and is the Medical Director of Pacific Movement Disorders Center at Providence Saint John’s Health Center and Providence Little Company of Mary Medical Center in Torrance. Dr. Petrossian’s clinical interests and expertise are in movement disorders such as Parkinson’s disease (PD), tremor, dystonia, gait disorders, ataxia, myoclonus, blepharospasm, hemifacial spasm, spasticity, and tics; as well as Parkinson’s-related conditions such as Dementia with Lewy Bodies, progressive supranuclear palsy, and multiple system atrophy. Dr. Petrossian’s focus is on a multidisciplinary approach to these movement disorders, including appropriate diagnosis utilizing the latest imaging techniques such as MRI, PET and DaTscan; optimization of medical treatment; Botox injections; and coordination with physical therapists and speech therapists. She assesses patients for deep brain stimulation surgery and creates a custom-tailored program of DBS settings. She is a member of the American Academy of Neurology and Movement Disorders Society.
Dr. Melita Petrossian is a Harvard-trained Neurologist and Movement Disorders specialist and is the Medical Director of Pacific Movement Disorders Center at Providence Saint John’s Health Center and Providence Little Company of Mary Medical Center in Torrance. Dr. Petrossian’s clinical interests and expertise are in movement disorders such as Parkinson’s disease (PD), tremor, dystonia, gait disorders, ataxia, myoclonus, blepharospasm, hemifacial spasm, spasticity, and tics; as well as Parkinson’s-related conditions such as Dementia with Lewy Bodies, progressive supranuclear palsy, and multiple system atrophy. Dr. Petrossian’s focus is on a multidisciplinary approach to these movement disorders, including appropriate diagnosis utilizing the latest imaging techniques such as MRI, PET and DaTscan; optimization of medical treatment; Botox injections; and coordination with physical therapists and speech therapists. She assesses patients for deep brain stimulation surgery and creates a custom-tailored program of DBS settings. She is a member of the American Academy of Neurology and Movement Disorders Society.
About the Author

Melita Petrossian
Melita Petrossian, MD, is Director of Pacific Movement Disorders Center and is a fellowship-trained neurologist with clinical interests and expertise in movement disorders such as Parkinson’s disease, essential tremor, dystonia, gait disorders, ataxia, myoclonus, blepharospasm, hemifacial spasm, Meige syndrome, spasticity, tics, and Tourette’s syndrome. She also specializes in Parkinson’s-related conditions such as Dementia with Lewy Bodies, progressive supranuclear palsy, multiple system atrophy, corticobasal degeneration, primary freezing of gait, and Parkinson’s disease dementia.
Last updated: April 19th, 2018