
Parkinson’s Disease: Understanding the Gut-Brain Connection (Part 3)
by Melita Petrossian
In part 3 of this 3-part blog series, I examine the research and theories about the gut’s connection to the brain and Parkinson’s disease.
What is the current understanding of the gut-brain connection in Parkinson’s disease (PD)?
Alpha-synuclein deposits can be seen in the gut prior to development of motor symptoms of Parkinson’s disease (PD), up to 20 years prior to diagnosis. Alpha-synuclein refers to the abnormal protein that is also seen in the brains of patients with PD. There can also be PD-type pathology (abnormalities) found in the parasympathetic neurons of the vagus and sacral nerves. These nerves connect the brain to the visceral organs and are involved in the autonomic (i.e., automatic, subconscious) nervous system.
 PD patients and normal control subjects have different types of gut bacteria, known as the gut microbiome. This is a new area of medical research where a less diverse (more limited) gut microbiome has been associated with autoimmune diseases and chronic inflammatory diseases. However, because these are associations, it’s important to remember that no causative conclusion can be drawn. That is, the diseases may be limiting the gut microbiome and not the other way around, or there may be a third factor (such as the use of antibiotics) that explains both the disease and the limited gut microbiome.
PD patients and normal control subjects have different types of gut bacteria, known as the gut microbiome. This is a new area of medical research where a less diverse (more limited) gut microbiome has been associated with autoimmune diseases and chronic inflammatory diseases. However, because these are associations, it’s important to remember that no causative conclusion can be drawn. That is, the diseases may be limiting the gut microbiome and not the other way around, or there may be a third factor (such as the use of antibiotics) that explains both the disease and the limited gut microbiome.
In addition to the presence or absence of PD, there appears to be different gut microbiome signatures in patients who are taking COMT inhibitors (such as entacapone (Comtan, Stalevo)) and anticholinergics (such as Artane (trihexyphenidyl)).
How could bacterial diversity in the gut affect the development of PD? There are a few theories, one of which involves short-chain fatty acids (SCFA), which are made by bacteria in the gut by breaking down dietary fiber, notably Lachnospiraceae. Two studies showed reduced levels of Lachnospiraceae in PD, which is consistent with SCFA depletion. SCFA deficiency is an attractive hypothesis for PD because it could potentially explain inflammation and microglial activation in the brain and gastrointestinal features of the disease (such as constipation), but it is not the whole picture. On the other hand, depletion of SCFA and SCFA-producing organisms has been observed in diverse disorders, which suggests that SCFA deficiency may be a common consequence of illness rather than a specific cause or even a biomarker for PD. Further supporting the microbiome theory is information that one environmental factor associated with PD, namely pesticide exposure, is known to reduce bacterial diversity in the gut.

One study looked more directly at what effect the gut microbiome could have by studying a mouse model of PD, specifically mice that overproduce alpha-synuclein and exhibit symptoms of PD. They divided the mice into two groups: one had the normal diverse gut bacteria and the other were bred in a completely sterile environment and therefore lacked gut bacteria. The researchers had both groups of mice perform several tasks to measure their motor skills, such as running on treadmills, crossing a beam, and descending from a pole. The germ-free mice performed significantly better than the mice with a complete microbiome. Rather than suggesting that we eradicate ourselves of gut bacteria, this simply tells us that gut bacteria regulate, and are even required for, the symptoms of PD.
The mechanism by which gut bacteria contribute to the symptoms of PD is hypothesized to be the formation of SCFA. The researchers then went on to inject SCFA into the guts of germ-free mice and showed motor disabilities consistent with parkinsonism and alpha-synuclein aggregation in the brain. This suggests that it is the SCFA that contributes to conversion of alpha-synuclein into disease-forming deposits. However, this theory appears to be contradicted by the reports of PD patients having lower SCFA levels in the gut. More likely, the apparent contradiction highlights the complexities of the gut microbiome and individual molecules thought to play a role in PD development.
Another theory of how conditions related to the gut could contribute to the development of PD involves excessive stimulation of the innate immune system due to abnormal gut bacterial profiles and/or small intestinal bacterial overgrowth (which is more common in PD patients than in control populations), which may lead to increased intestinal permeability, thus producing systemic and/or CNS inflammation. (Though PD is not an autoimmune condition like multiple sclerosis, there are associations of inflammatory states with PD, as well as epidemiologic evidence that use of anti-inflammatory medications, namely ibuprofen, were associated with a lower risk of development of PD. Please note this does not mean that patients with PD should take ibuprofen or other anti-inflammatory medications; there is no evidence this would help.)
Connecting the gut to the brain in PD, another piece of evidence is the frequency of finding Lewy bodies (the abnormal collections of protein in nerve cells seen in PD and Dementia with Lewy Bodies) along the gastrointestinal tract. The highest frequency is found in the submandibular gland (located under the jaw, producing saliva) and lower esophagus. Lower frequencies are found in the stomach, small intestine, colon and rectum. This represents a gradient in which the locations closer to the brain have a higher frequency of Lewy bodies. This corresponds to Braak’s hypothesis which suggests that PD-related pathology starts in the enteric (gut-related) nervous system and spreads retrogradely (backwards along the course of nerves) to the brain via the vagus nerve. Alpha-synuclein would then enter a part of the brainstem, then spread to the substantia nigra, where motor systems would then be affected.
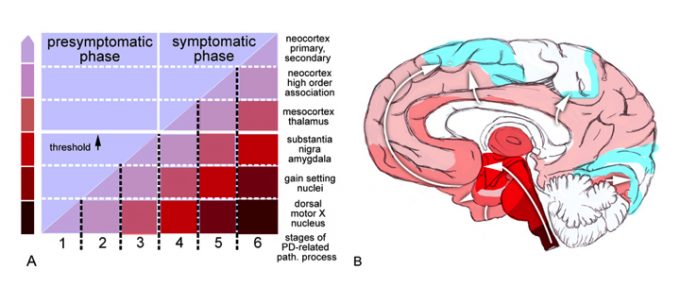
What does this mean in terms of prevention of PD or prevention of progression for PD?
There is a lot of promising research ongoing about utilizing saliva, salivary glands, and the gut (stool samples) to identify biomarkers for PD. These may serve to identify people at risk of developing PD, and/or help determine markers of progression for patients with PD. This potential information could be of huge importance in that many earlier-stage patients may be assessed for risk and more timely assessments of disease progression could be made, allowing for earlier drug development rather than relying on clinical progression treatment, DaTscan findings or pathology findings, which take years.
There is no evidence to support the use of probiotics or pre-biotics although this is an area of early research for the management of gut-related symptoms such as constipation. Other groups are considering studying the use of fecal transplantation, to transfer live bacteria from a healthy person’s gut to another.
Studies for disease prevention and prevention of progression in Parkinson’s disease will take larger and longer studies which requires ongoing funding.

Programs such as the American Parkinson’s Disease Association’s (APDA) Optimism Walk aim to raise awareness and money for research.
Register online today for the 2017 Los Angeles area walk!
Stop by our booth to meet Dr. JP Langevin, Director of Restorative Neurosurgery, and our new Nurse Practitioner, Giselle Tamula.
Saturday, April 22, in Palos Verdes
apdaparkinson.org/CAwalk or call 310-486-0153
Read more about Parkinson’s disease and the gut in Part 1 and Part 2 of this 3-part blog series.
About the Author

Melita Petrossian
Melita Petrossian, MD, is Director of Pacific Movement Disorders Center and is a fellowship-trained neurologist with clinical interests and expertise in movement disorders such as Parkinson’s disease, essential tremor, dystonia, gait disorders, ataxia, myoclonus, blepharospasm, hemifacial spasm, Meige syndrome, spasticity, tics, and Tourette’s syndrome. She also specializes in Parkinson’s-related conditions such as Dementia with Lewy Bodies, progressive supranuclear palsy, multiple system atrophy, corticobasal degeneration, primary freezing of gait, and Parkinson’s disease dementia.
Last updated: April 2nd, 2020