Inspire Surgical Implant
What is the Inspire Surgical Implant for Obstructive Sleep Apnea?
Surgery to implant the Inspire device for obstructive sleep apnea (OSA) offers an alternative for individuals who struggle with CPAP machines. The Inspire device and CPAP machine are both treatments for obstructive sleep apnea (OSA), but they function differently. CPAP delivers a continuous stream of air through a mask, keeping the airway open during sleep. While effective, many patients find the mask uncomfortable and challenging to use consistently.
In contrast, the Inspire device is a surgically implanted system that stimulates the hypoglossal nerve to maintain an open and unobstructed airway. This method does not involve a mask or airflow, making it a more comfortable alternative for those who struggle with CPAP compliance.
Patient outcomes with the Inspire device implant have been promising. 90% of bed partners report no snoring or soft snoring, and there is a 79% reduction in sleep apnea events. Remarkably, 94% of patients report that they prefer the implant over CPAP and would recommend it to others.
How Does Inspire Work?
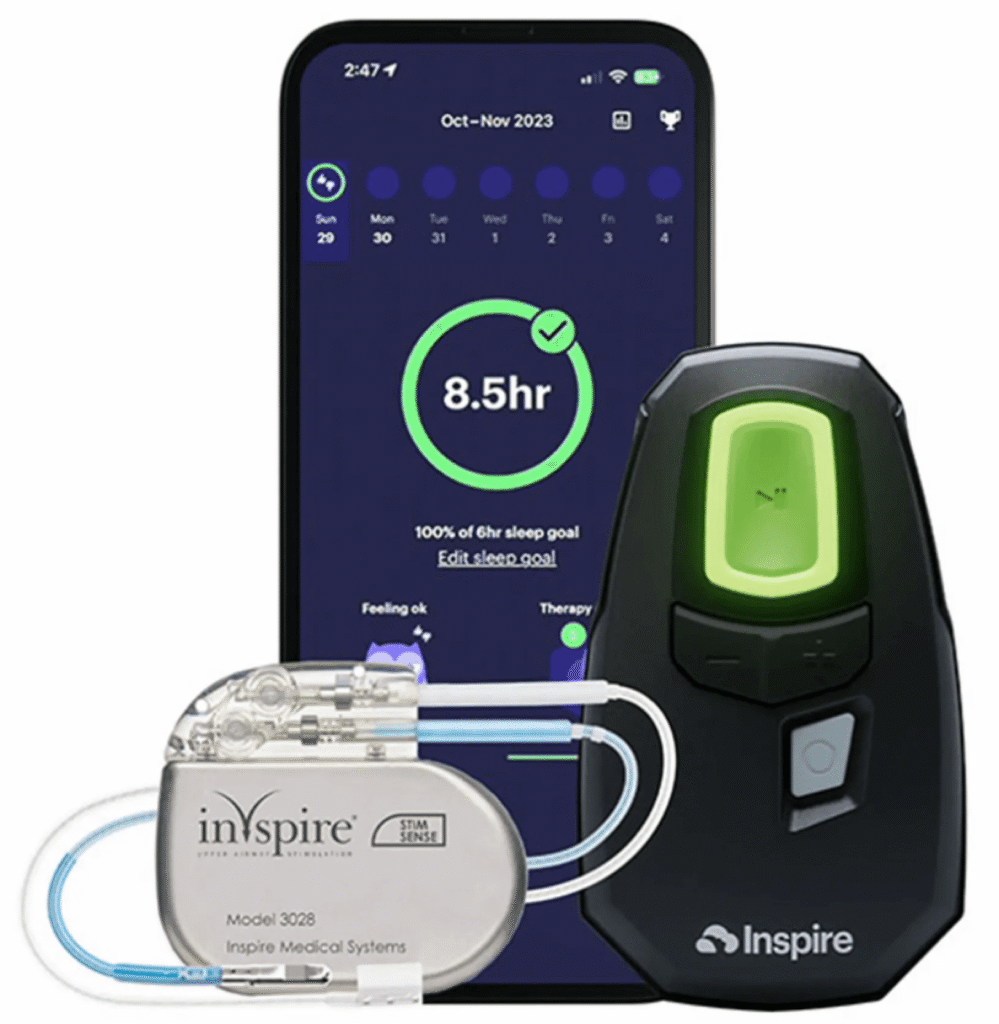
This minimally invasive surgery involves implanting a small device that works by stimulating the hypoglossal nerve, which controls tongue movement. The device helps keep the airway open during sleep, reducing apneas and improving sleep quality. Unlike traditional surgeries for OSA, Inspire therapy doesn’t involve removing tissue or altering the airway structure. Instead, it uses a gentle, rhythmic pulse to prevent airway collapse and eliminate or improve sleep apnea.
Patients activate the Inspire device each night using a handheld remote before sleeping. This targeted approach can be particularly beneficial for those who cannot tolerate CPAP masks and seek a more manageable long-term solution.

Benefits of Inspire Therapy
Inspire therapy offers significant benefits for those with obstructive sleep apnea (OSA) who struggle with CPAP machines. By providing targeted stimulation to the hypoglossal nerve during sleep, Inspire improves sleep quality and significantly reduces sleep apnea events. Patients often experience fewer interruptions in their sleep cycles, leading to more restful and restorative sleep.
Compared to CPAP, Inspire surgery tends to have increased compliance rates. CPAP requires wearing a mask throughout the night, which can be uncomfortable and difficult for many people. Inspire, on the other hand, involves a minimally invasive surgery for OSA to implant the device, which patients activate using a handheld remote before sleeping. This system eliminates the need for cumbersome masks and hoses, making it a more comfortable and convenient option. Consequently, patients are more likely to adhere to Inspire therapy, leading to consistent and effective management of their sleep apnea. Discuss Inspire therapy with your healthcare provider to see if it is suitable for you.
Who is a Good Candidate for Inspire Surgery?
A good candidate for Inspire surgery typically includes individuals with moderate to severe obstructive sleep apnea (OSA) who have difficulty tolerating or complying with continuous positive airway pressure (CPAP) therapy. Specifically, candidates for Inspire therapy should meet the following criteria:
- Diagnosis of Moderate to Severe OSA: This is typically determined by a sleep study that measures the apnea-hypopnea index (AHI). Candidates usually have an AHI between 15 and 65 events per hour.
- Inability to Tolerate CPAP Therapy: Patients who have tried CPAP but find it uncomfortable, cumbersome, or ineffective are ideal candidates.
- Body Mass Index (BMI): A BMI of less than 32 is often required, though this can vary slightly based on specific guidelines and individual assessments.
- No Complete Concentric Collapse (CCC) at the Soft Palate: This is determined via a sleep endoscopy procedure. Patients with CCC may not benefit from Inspire surgery.
- Age and General Health: Typically, candidates are over 22 years old and in generally good health to undergo a surgical procedure.
Learn More About Inspire Surgical Implant
Inspire is an FDA‑approved implantable device for treating moderate to severe obstructive sleep apnea in adults who have difficulty tolerating CPAP therapy. It works by delivering gentle electrical pulses to the nerve that controls tongue and airway muscles, helping keep the airway open during sleep.
Unlike CPAP, which uses a mask and pressurized air to keep the airway open, Inspire works inside the body. You activate it each night with a small handheld remote, and it helps maintain an open airway without masks, hoses, or forced air pressure.
Inspire is generally recommended for adults with moderate to severe OSA who:
- Have been unable to tolerate or comply with CPAP therapy
- Have an apnea‑hypopnea index (AHI) within an eligible range
- Have a body mass index (BMI) below a specified cutoff
- Have <25% central or mixed apnea events
- Undergo drug‑induced sleep endoscopy confirming airway collapse pattern suitable for the device
Inspire is placed during a minimally invasive outpatient surgery that typically takes a couple of hours under general anesthesia. The device is implanted in the chest with leads to the nerve that controls tongue movement. After healing, it’s activated and carefully titrated to each patient’s needs.
After surgery, there is a healing period. The device is usually activated about 6 weeks post‑implantation, and therapy settings are adjusted over the following weeks to optimize comfort and effectiveness. A sleep study is often used to assess improvement.
Clinical experience shows that Inspire can significantly reduce apnea events, often improving sleep quality and daytime alertness. Many patients and their partners report reduction in snoring and fewer breathing interruptions. However, results vary, and it may not completely eliminate OSA for every patient.
As with any surgical implant, there are risks including infection, pain, swelling, or nerve‑related effects. Once active, some patients may experience mouth dryness, tongue discomfort, or stimulation sensations. Most side effects are mild or can be managed with adjustments, though rare complications can occur.
Yes. If needed, the device can be surgically removed. Because it’s implanted under the skin with leads to the nerve, removal is more involved than the initial placement and should be discussed with your surgeon.
Certain types of medical imaging (e.g., some MRI protocols) may be restricted with an implanted device. Your physician or radiologist should be consulted before scheduling imaging.
Many insurance plans, including Medicare in many cases, provide coverage for Inspire therapy for eligible patients. Coverage varies by plan and individual patient criteria, so verifying with your insurer is recommended.
For many patients who meet eligibility criteria and commit to follow‑up care, Inspire offers a long‑term alternative to CPAP with sustained improvements in sleep quality and breathing during sleep. Regular follow‑up with a sleep specialist helps maintain effectiveness and monitor device function.
Resources
Get Expert Care from Leading Specialists
Our Inspire surgery implant experts can be reached at 11645 Wilshire Blvd., #600, Los Angeles, CA 90025. 310-477-5558
Written and reviewed by:
The Pacific Neuroscience medical and editorial team
We are a highly specialized team of medical professionals with extensive neurological and cranial disorder knowledge, expertise and writing experience.
Last Updated:
