
Pituitary Adenoma
What is a Pituitary Adenoma?
A pituitary adenoma is a benign (noncancerous) growth on your pituitary gland, a small gland located at the base of the brain. The pituitary gland is responsible for producing hormones that regulate various bodily functions, including growth, metabolism, reproduction, and stress response. Pituitary adenomas are the third most common intracranial tumor and arise from the pituitary gland.
Pituitary adenomas can be classified based on their size and hormone-producing activity. Microadenomas are small tumors that are less than 1 centimeter in size, while macroadenomas are larger tumors that are more than 1 centimeter in size. Some pituitary tumors can also produce excess hormones, leading to hormonal imbalances and associated symptoms.
The exact cause of pituitary adenomas is not known, but genetic factors and hormonal imbalances may play a role. Some pituitary adenomas may also be associated with underlying medical conditions, such as multiple endocrine neoplasia type 1 (MEN1) or Carney complex.
Types of Pituitary Adenomas
Pituitary adenomas are tumors that develop in the pituitary gland, a small gland located at the base of the brain. These tumors can be categorized based on their size, behavior, and the hormones they produce. The main types of pituitary adenomas include:
Prolactinomas
Tumors that produce excessive prolactin hormone.
Growth hormone-secreting adenomas
Tumors that produce excessive growth hormone.
ACTH-secreting adenomas
Tumors that produce excessive adrenocorticotropic hormone.
Non-functioning adenomas
Tumors that do not produce excessive hormones but may cause symptoms due to their size or location.
Symptoms and Impact on Hormone Production
Pituitary adenomas can cause a range of symptoms depending on their type, size, and the hormones they produce. These symptoms may include headaches, vision problems, hormonal imbalances, fatigue, and changes in menstrual cycles. The impact on hormone production can lead to various endocrine disorders, affecting growth, reproduction, metabolism, and other bodily functions.
Diagnosis and Treatment Options
Diagnosing pituitary adenomas involves a comprehensive evaluation, including medical history review, physical examination, hormone level testing, and imaging studies such as MRI or CT scans. Our team of specialists utilizes advanced diagnostic techniques to accurately identify and classify pituitary tumors, enabling personalized treatment plans.
Treatment Approaches for Pituitary Adenomas
The treatment options for pituitary adenomas depend on several factors, including the tumor type, size, symptoms, and the patient’s overall health. Treatment modalities may include:
- Medication: Certain pituitary adenomas can be effectively managed with medication to regulate hormone levels and control tumor growth.
- Surgery: Surgical intervention may be recommended for larger or hormone-secreting tumors. At Pacific Neuroscience Institute, we specialize in minimally invasive endoscopic endonasal surgery, which offers excellent outcomes with reduced risks and faster recovery.
- Radiation therapy: In some cases, radiation therapy may be used to target and shrink pituitary tumors.
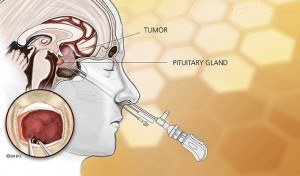
Surgical removal via the endoscopic endonasal approach is the treatment of choice for most patients with asymptomatic pituitary adenoma (with the exception of prolactinomas).
Given major technical advances over the last two decades in high-definition endoscopy, surgical navigation techniques and more effective skull base closure methods, pituitary surgery is safer than ever in experienced hands.
Pituitary Adenoma Neurosurgeons and Specialists
At the Pacific Pituitary Disorders Center in Santa Monica, California, we have one of the world’s largest experiences in endoscopic endonasal transsphenoidal surgery for all types of pituitary adenomas including acromegaly, Cushing’s disease, prolactinomas, non-functional adenomas, pituitary apoplexy and residual or recurrent adenomas.
Since 2010, our PNI neurosurgeons and ENT surgeons crossed a milestone of operating on over 500 patients with a pituitary adenoma with a fully endoscopic endonasal approach, and our median hospital length of stay has dropped to just one day.
By incorporating leading-edge technology and instrumentation with proven surgical experience of over 2,000 endonasal surgeries, our pituitary tumor specialists led by neurosurgeons Dr. Daniel Kelly, Garni Barkhoudarian and Walavan Sivakumar, and ENT/Otolaryngologist Dr. Chester Griffiths, make pituitary surgery safer, less invasive and more effective.
About Pituitary Adenomas
Pituitary adenomas (tumors) arise from the pituitary gland and are by far the most common growth associated with the gland; they account for 15% of primary brain tumors and are the third most common intracranial tumor after meningiomas and gliomas.
The great majority (over 90%) of pituitary adenomas are benign slow-growing tumors, approximately 5-10% are somewhat more aggressive (atypical) and less than 1% qualify as pituitary carcinoma.
From autopsy studies and MRIs of normal individuals, it is known that 10-20% of the general population has a pituitary adenoma. Most of these tumors remain small and do not cause significant harm or symptoms; however many do progress and grow to cause hormonal and neurological problems.
Types of Pituitary Adenomas
Pituitary adenomas are classified by size and whether they produce pituitary hormones; microadenomas are less than 1 cm in diameter and macroadenomas are over 1 cm in diameter.
Endocrine-active adenomas
Adenomas that make excess hormones (endocrine-active adenomas) include prolactin-secreting adenomas known as prolactinomas, adrenocorticotropic hormone (ACTH) secreting adenomas causing Cushing’s disease, growth hormone (GH) secreting adenomas causing acromegaly, and thyroid stimulating hormone (TSH) secreting adenomas causing hyperthyroidism.
Endocrine-inactive Adenoma
Adenomas that do not make excess hormones are called endocrine-inactive or Non-Functional Adenomas. Most adenomas are not genetically inherited; cases of familial pituitary tumors are rare. Multiple Endocrine Neoplasia (MEN) type I accounts for less than 5% of cases of pituitary adenomas. This autosomal dominant condition is characterized by multiple and sometimes simultaneous tumors of the pituitary, pancreas and parathyroid glands. Pituitary adenomas develop in 25% of patients with MEN I.
Symptoms of Pituitary Adenomas
Symptoms of a pituitary adenoma can vary depending on the size and hormone-producing activity of the tumor. Common symptoms may include headaches, vision problems, fatigue, weight gain or loss, menstrual irregularities, decreased libido, and infertility. Pituitary adenomas may cause problems because of hormonal hyper-secretion. In some cases, pituitary adenomas may also lead to excessive thirst or urination, high blood pressure, or mood changes.
Hormonal hypersecretion
The three most common hormonally active adenomas are prolactinomas, GH-secreting tumors causing acromegaly, and ACTH-secreting tumors causing Cushing’s disease. Thyroid stimulating hormone (TSH) tumors are relatively rare.
Pituitary hormonal deficiency (Hypopitutiarism)
(Hypopituitarism) This problem typically occurs only in larger tumors (macroadenomas) and results from compression and damage to the normal pituitary gland from the enlarging adenoma.
Manifestations may include:
- Hypogonadism – sexual dysfunction, loss of libido, and impotence
- Hypothyroidism – fatigue, weakness, weight gain, coarse dry hair and dry skin, cold intolerance, depression
- Adrenal insufficiency – fatigue, weakness, loss of appetite, dizziness, nausea and vomiting
- Growth failure – in children and adolescents
- Hyperprolactinemia – due to “stalk effect”. This is seen in diseases within or near the pituitary gland and stalk. Interfering with the delivery of dopamine, a neuron-transmitter, from the hypothalamus to the prolactin secreting cells of the pituitary gland), which can result in hypogonadism and its associated problems. Rarely posterior pituitary gland damage occurs with diabetes insipidus, which is caused by the inability of the kidneys to conserve water, leading to frequent urination and thirst. Learn more about hypopituitarism
Neurological problems
Larger pituitary macroadenomas often cause loss of visual acuity or peripheral vision (termed bitemporal hemianopsia) from pressure on the optic nerves and optic chiasm which is directly above the pituitary gland. Larger macroadenomas especially those that have hemorrhaged (apoplexy) may also cause double vision.
Headache
Patients with macroadenomas often have frontal, forehead and temporal area headaches. Pituitary adenoma removal often results in headache resolution.
Bleeding (pituitary apoplexy)
This condition develops over hours to several days from hemorrhage and/or infarction of pituitary adenoma (typically a macroadenoma or a Rathke’s cleft cyst).
Symptoms may include:
- Headache
- Nausea
- Visual loss
- Double vision
- Confusion
Most patients have undiagnosed hormone insufficiency prior to the apoplectic event. Pituitary apoplexy is best confirmed by MRI. A head CT will also show areas of bleeding or a mass in the sella in the majority of cases. Other conditions that can mimic pituitary apoplexy are a ruptured aneurysm, meningitis, a stroke, intracerebral hemorrhage and migraine headache.
The treatment for most patients with pituitary apoplexy is urgent endoscopic endonasal surgery and intravenous administration of the stress hormone cortisol.
Diagnosis of Pituitary Adenomas
Pituitary adenomas are best diagnosed by imaging studies and hormonal testing.
Imaging
The imaging study of choice is an MRI of the pituitary gland without and with gadolinium (a contrast agent). A brain MRI or CT scan will also reveal most pituitary macroadenomas but may not reveal smaller microadenomas.
In a minority of patients it may be difficult to distinguish an adenoma of the pituitary from other masses which may include:
- Craniopharyngioma
- Rathke’s Cleft cyst
- Meningioma
- Hypophysitis (pituitary inflammation)
- Glioma of the suprasellar region
- Metastatic tumor
- Cordoma
Pituitary Hormonal Testing
Evaluation and interpretation of the pituitary gland function either for hormonal deficits or inappropriate hormonal secretion is performed in our center, under the supervision of our pituitary neuroendocrinologist Dr. Katherine Araque.
Our team recognizes that one size does not fit all. Consequently, pituitary hormonal testing is individualized to the needs of our patients.
Our center can be easily perform these types of blood level measurements:
- ACTH
- Serum cortisol
- TSH
- Free T4
- Total T3 (thyroid function)
- LH (luteinizing hormone)
- FSH (follicle-stimulating hormone)
- Estradiol in women
- Free and total testosterone in men
- GH (growth hormone)
- IGF-1 (insulin-like growth factor)
- Prolactin
- Macroprolactin
Additionally, patients can have access to various diagnostic tools including radiologic images with selected pituitary protocols, noninvasive and invasive dynamic endocrinology testing.
Neuro-Ophthalmological Evaluation
Patients with visual complaints or those whose tumors that contact the optic nerves or optic chiasm should receive a full ophthalmological evaluation. An evaluation with our neuro-ophthalmologist, Dr. Howard Krauss, should include acuity (vision quality) testing of each eye and formal visual field testing to determine if there is loss of peripheral vision.
Treatment
Treatment options for pituitary adenomas depend on the size and hormone-producing activity of the tumor, as well as the associated symptoms. In some cases, close monitoring and observation may be recommended for small, non-functioning tumors that are not causing any symptoms. Treatment options for larger or hormone-producing tumors may include medications to reduce hormone levels, surgery to remove the tumor, or radiation therapy.
Pituitary Adenoma (Tumor) Surgery
Endoscopic Endonasal Surgery
The first-line treatment for all pituitary adenomas (tumors) except prolactinomas (as discussed below), as well as Rathke’s Cleft Cysts (RCCs) and most craniopharyngiomas is endoscopic endonasal transsphenoidal surgery. Because of improved tumor visualization and tumor removal rates, the endoscopic endonasal approach has become the preferred method for removal of pituitary tumors, RCCs, as well as the great majority of craniopharyngiomas, clival chordomas and many midline meningiomas.
Surgical success rates (complete tumor removal and normalization of hormonal hypersecretion) are generally quite high (>80-90%) with smaller and non-invasive pituitary adenomas, but are lower for large or invasive macroadenomas (40-70%).
Major surgical complications such as vision loss, bleeding, stroke, cerebrospinal fluid leak and meningitis are low when performed by experienced endonasal transsphenoidal neurosurgeons who often work collaboratively with a Head & Neck surgeon. The success rates of surgery are described further in the sections below for specific tumor types.
Transcranial Surgery
A craniotomy (opening in the skull from above to reach the pituitary region) is required in less than 1% of patients who require surgery for a pituitary adenoma.
Medical & Radiation Therapies
See specific pituitary adenoma subtypes:
Hormone Replacement Therapy
Many patients with pituitary tumors will develop varying degrees of hypopituitarism (pituitary gland deficiency) either as a result of the tumor impact on the gland or sometimes as a result of surgery or other treatments. While many patients can have some degree of gland recovery, many will ultimately require short-term or long-term hormone replacement therapy. Our highly experienced pituitary endocrinologists, Drs. Pejman Cohan and Katherine Araque can provide tailored hormone replacement therapy including for low testosterone, low estrogen, growth hormone deficiency, hypothyroidism, low cortisol (adrenal insufficiency) and diabetes insipidus (anti-diuretic hormone deficiency).
For prolactinomas that are typically treated with medical therapy, we have highly experienced endocrinologists to provide this therapy. For patients with adenomas that cannot be completely treated with surgery and/or medical therapy, our experienced radiation oncologists experts can deliver focused radiosurgery or radiotherapy to tumors, halting their growth.
We also have clinical trials available using novel medical therapies for patients with persistent acromegaly and Cushing’s disease who failed prior treatments.
Is a brain tumor, pituitary lesion and a pituitary tumor the same thing?
A pituitary tumor is considered a type of brain tumor. Pituitary lesions may also include cysts and inflammatory conditions that may mimic tumors.
What pituitary tumor treatment do you recommend?
One specific type of pituitary tumor, a prolactinoma (pituitary adenoma that secretes the hormone prolactin), can be very successfully treated with medications (cabergoline or other dopamine agonist).
In all other patients who have symptomatic pituitary adenomas or have a growing tumor, especially in patients with Cushing’s disease or acromegaly, or other pituitary conditions such as central hyperthyroidism, craniopharyngioma and chordoma, surgical resection is best.
We recommend an endoscopic endonasal surgical removal of the tumor by an experienced team, ideally consisting of both a neurosurgeon and ENT surgeon.
I’m worried about my pituitary tumor diagnosis. Is pituitary tumor surgery dangerous?
There are always risks with any surgery. Though not very common, there are certainly serious complications that can occur during or after pituitary tumor surgery. These range from sinus infection, bleeding and hormone dysfunction to meningitis, vision loss, stroke and even death. Thankfully, the risks of these major complications are low (1-3%).
How long does pituitary tumor surgery take?
The duration of pituitary tumor surgery varies based on the tumor type, specific location, invasiveness and goals of surgery. The typical operation is around 3 hours, but can be upwards of 8-10 hours for rare tumors.
How common are pituitary tumors?
Pituitary tumors and cysts are very common, being identified in about one in six people. However, those that need surgery are much less common, affecting approximately 1/10,000 people annually.
How fast do pituitary tumors grow?
Most pituitary tumors are slow growing, approximately 1-3mm/year.
How often do pituitary tumors grow back?
This depends on the tumor type and pathological features. Most pituitary tumors have a recurrence rate of about 10% over 10 years. Craniopharyngiomas, chordomas and Cushing’s disease tend to have faster recurrence rates.
Are there pituitary disorders support groups that I can go to?
Yes, the Pacific Pituitary Disorders Center’s patient support group is the longest running group in the United States. Patient Advocate, Sharmyn McGraw has a vast network of resources available to patients seeking community support.
What are the psychiatric symptoms of pituitary adenoma?
Pituitary adenomas, which are benign tumors of the pituitary gland, can lead to a variety of psychiatric symptoms due to hormonal imbalances or pressure on surrounding brain structures. Common psychiatric symptoms include:
- Mood Disorders: Depression and anxiety are frequently observed. Patients may experience persistent sadness, loss of interest in activities, or excessive worry and fear.
- Cognitive Changes: Difficulties with concentration, memory, and decision-making can occur. Some patients might report brain fog or decreased mental sharpness.
- Personality Changes: Irritability, emotional instability, and changes in behavior or personality may develop.
- Psychosis: Although rare, some individuals may experience hallucinations or delusions.
- Sleep Disturbances: Insomnia or excessive sleepiness can result from hormonal imbalances affecting sleep regulation.
- Sexual Dysfunction: Changes in libido, erectile dysfunction in men, and menstrual irregularities in women can impact psychological well-being.
- Appetite Changes: Abnormalities in appetite and weight gain or loss might be present, contributing to mood and anxiety issues.
These symptoms arise due to the tumor’s impact on hormone production or compression of nearby brain areas, affecting emotional and cognitive functions. Early diagnosis and treatment are crucial for managing both the physical and psychiatric effects of pituitary tumors.
How long is the hospital stay after pituitary tumor surgery?
The hospital stay after pituitary tumor surgery at PNI is typically 1-2 days, depending on the patient’s health, surgery complexity, and complications. Initially, patients are monitored in an ICU or high-dependency unit for the first 24 hours. Close monitoring for complications such as cerebrospinal fluid leaks, infections, or hormonal imbalances is essential. Before discharge, endocrine evaluations, imaging studies, and assessments of mobility and basic activities are conducted. Patients are discharged once stable and mobile, with follow-up outpatient visits for ongoing monitoring and management of any long-term effects.
Doctors and Specialists Who Treat Pituitary Adenomas
Pituitary tumors and related hormonal disorders are complex in their diagnosis and treatment & warrant a team approach of experienced specialists to achieve optimal outcomes.
We have assembled a highly experienced team of experts who work closely together on your behalf. From diagnosis to treatment, as well as education and support, we are here for you.













