DBS for Parkinson’s Disease
Deep Brain Stimulation (DBS) can complement an optimized medication regimen for patients with Parkinson’s disease.
“On-Off” Time in Parkinson’s Disease (PD)
Unfortunately, at some point along the course of the diagnosis of Parkinson’s disease (PD), the brain no longer responds consistently to the medications. The effective window (known as ON time) is shorter, and thus there is more OFF time (when the patient feels slow, sluggish, or frozen because the medications haven’t kicked in yet or have worn off too soon).
Motor Complications in Parkinson’s Disease
In addition, there are increased dyskinesias (involuntary movements), which refer to abnormal involuntary movements (not including tremor). ON/OFF fluctuations and dyskinesias are known collectively as motor complications.
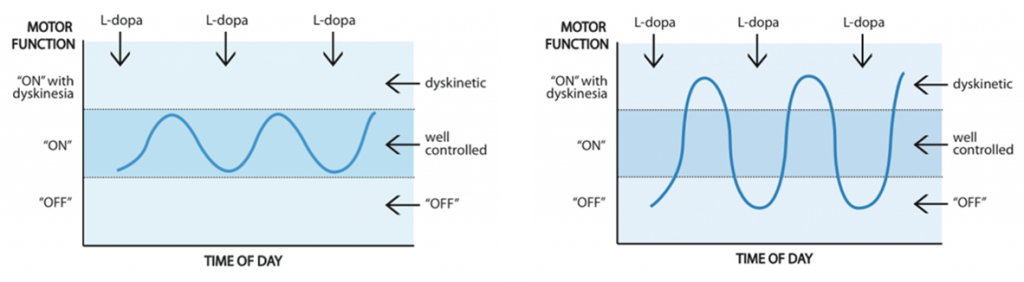
Whereas levodopa concentration fluctuations can contribute to unpredictable shifts between ON and OFF time, Deep Brain Stimulation remains constant through the day, meaning that there is consistent efficacy throughout the day.
Deep Brain Stimulation for Parkinson’s Disease Treatment
Patients note a significant improvement in ON time – meaning an addition of 5 good hours of productivity during the day. Deep Brain Stimulation (DBS) helps free patients from the worry of whether their medication is going to kick in at the right time for them to drive to work, participate in a meeting, have a meal, or pay a visit to a friend. This improves quality of life dramatically.
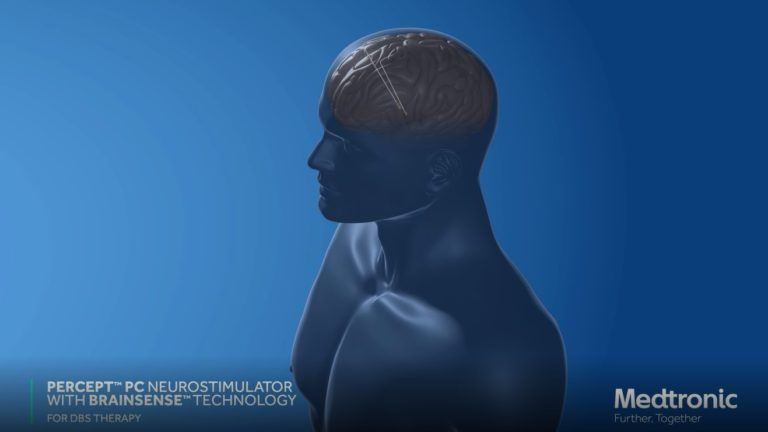
For many patients with Parkinson’s disease who continue to experience challenging symptoms, a state-of-the-art technology introduced in 2020 at Pacific Neuroscience Institute can provide comfort and relief.
The DBS surgically-implanted device called Percept™ PC Neurostimulator with BrainSense™ technology detects patient-specific brain signals and provides feedback to optimize therapy for patients with Parkinson’s disease. Benefits include improvements in tremors, rigidity, slowness of movement and overall quality of life.
Our movement disorders neurology and restorative neurosurgery specialists provide DBS expertise and experience. The first in Providence health system to use Percept™ PC with BrainSense™, this breakthrough in monitoring and treatment technology can be considered for patients who need to take their current medications too frequently, or who can no longer tolerate their current treatment regimen. This DBS system is available for new patients as well as for updating older implanted DBS systems.
Patients receive a comprehensive and personalized medical and treatment assessment to determine of DBS is an appropriate choice.
Deep Brain Stimulation and Dyskinesia
Deep Brain Stimulation (DBS) can also significantly reduce dyskinesia. DBS for Parkinson’s disease can be done in one of two sites: globus pallidus interna (GPi) or subthalamic nucleus (STN). When DBS targets STN, patients tend to have a reduction of their levodopa dose, in turn resulting in less dyskinesias. When DBS targets GPi, patients tend to require the same dose of levodopa, but the dyskinesias are reduced significantly even without any change in the dose of levodopa. The degree of sensitivity to medication (the degree of dyskinesia at low doses) as well as other factors will determine whether our team recommends GPi or STN implantation.
Levodopa and Deep Brain Stimulation for Parkinson’s Disease
Symptoms that do not respond to levodopa (such as balance problems) are unlikely to improve with Deep Brain Stimulation (DBS). Also if a patient with Parkinson’s Disease has no response (benefit) when taking adequate doses of levodopa, they are unlikely to experience any benefit from DBS. Sometimes patients would have a good response to levodopa, but they cannot tolerate the optimal dose.
This can be determined by doing a pre- and post-medication examination while the patient is in the clinic, giving a high dose of medication on an empty stomach. Patients who have good benefit to levodopa but cannot tolerate it may be good candidates for DBS. Patients should expect DBS to provide the same benefit as their best response to levodopa, but in a more consistent and reliable way. Please note that DBS is not a cure, and does not benefit imbalance, cognition, or freezing.
The only exception to the rule that DBS provides the same benefit as the best response to levodopa is tremor. Many patients with tremor-predominant PD find that while other motor symptoms (slowing, stiffness, shuffling, small handwriting) improve with medication, the tremor may not respond as much, or at all.
These patients do still enjoy significant benefit in tremor reduction with DBS, by about 75%.
By the numbers:
-
Since 1995, more than 160,000 patients with movement disorders have been treated with DBS worldwide.
-
DBS was FDA-approved for advanced PD in 2002.
-
In 2016, DBS was FDA-approved for use in earlier stages of PD: four years after onset and four months after the development of motor complications.
-
Medtronic’s Percept PC™ with BrainSense™ technology launched in 2020. It detects patient-specific brain signals and continuously gathers feedback to optimize therapy.
Who is a Candidate for DBS?
- Good initial response to levodopa
- On-off fluctuations (at least four months)
- Troubling dyskinesias
- Disabling tremor
- Absence of dementia or severe depression
- Normal MRI for age
- Good medical health
- Realistic expectations
Benefits of Deep Brain Stimulation for Parkinson’s Disease
- Improved “on-time” (gain of 5 hours/day on average)
- Reduced tremor (75% improved on average)
- Improved quality of life
- Reduced need for medication (25% reduction on average)
Risks and Side Effects of Deep Brain Stimulation Surgery
- Post-operative headache and pain are possible but typically resolve gradually a few weeks after the procedure.
- Neck pain is also possible in the short term. Infection of the leads, extension or battery is very low risk and our surgical team does their utmost to prevent this.
- Hemorrhage is extremely low risk but when it occurs may lead to stroke-like symptoms. Most frequently, hemorrhage does not result in permanent complications.
- Lead fracture or migration, also very low risk, may require repeat surgery.
- Problems with speech, language and mood; muscle tightness, slurred speech and vision symptoms can be related to the stimulation and thus can usually be reduced or eliminated by adjusting the stimulation.
Deep Brain Stimulation Doctors

Our expert restorative neurosurgeon Dr. JP Langevin has many years of experience with DBS. He was the first to implant and program PerceptTM PC with BrainSenseTM technology DBS system to treat patients with Parkinson’s disease among Providence hospitals, the 3rd largest healthcare system in the United States.
Contact Us:
Our movement disorders neurology and restorative neurosurgery team can be reached at 310-582-7433.
Additional Resources
Videos about Deep Brain Stimulation (DBS)
 What is Deep Brain Stimulation (DBS)? | Dr. Jean-Philippe Langevin
What is Deep Brain Stimulation (DBS)? | Dr. Jean-Philippe Langevin
 Charles’ Story | Parkinson’s Disease Treatment with DBS
Charles was living a full life when Parkinson’s disease hit. Although a progressive neurodegenerative disease affecting movement, there are many medical treatments available to help with symptom management. While Levadopa,…
Charles’ Story | Parkinson’s Disease Treatment with DBS
Charles was living a full life when Parkinson’s disease hit. Although a progressive neurodegenerative disease affecting movement, there are many medical treatments available to help with symptom management. While Levadopa,…
 Pacific Movement Disorders Center
Pacific Movement Disorders Center
 Meet Dr. JP Langevin
Meet Dr. JP Langevin
 DBS Off-On Demo: Parkinson’s Symptoms
DBS Off-On Demo: Parkinson’s Symptoms
 Percept PC™ with BrainSense™ Technology DBS system for Patients with Parkinson’s Disease
Percept PC™ with BrainSense™ Technology DBS system for Patients with Parkinson’s Disease

What is Deep Brain Stimulation (DBS)? | Dr. Jean-Philippe Langevin

Charles’ Story | Parkinson’s Treatment with DBS
Pacific Movement Disorders Center

Meet Dr. JP Langevin

DBS Off-On Demo: Parkinson’s Symptoms